
The basic principles of a hepatitis A vaccine schedule
CLICK to go back to FAQ menu
1. For a short e learning programme to reinforce all the below facts go to NUGGETS OF KNOWLEDGE – HEPATITIS A VACCINES
2. And an ARTICLE published in September 2025 entitled FREQUENTLY ASKED QUESTIONS ABOUT NHS TRAVEL IMMUNISATION. Thanks go to Independent Nurse for allowing me to share this information which covers the hepatitis A vaccine products and also the combined hepatitis A+B vaccines.
INTRODUCTION
Hepatitis A vaccine information has always caused many queries, but the general rules really make it quite easy to follow these days. This information applies to the MONOVALENT vaccines (ie single hepatitis A vaccines) discussed below.
There are 6 different hepatitis A vaccines at the current time as follows:
- GlaxoSmithKline supply Havrix Monodose, Havrix Junior Monodose
- MSD supply VAQTA adult and VAQTA paediatric
- Sanofi Pasteur supplies Avaxim and Avaxim Junior
A course of hepatitis A vaccine comprises 2 doses: a 1st dose and a second (or booster) dose. If you clicked on any of the named vaccines above you would be taken to the Electronic Medicines Compendium (emc) which provides details of the Summary of Product Characteristics (SmPC) or ‘license’ of a drug and the Patient Information Leaflet (PIL). The licenses of the hepatitis A vaccines vary as to when the booster dose would be given and the length of protection provided.
Immunisation against infectious disease which we call the GREEN BOOK is our national guidance in the UK for all vaccine preventable diseases. This also lays out the recommended schedules of vaccines. However, sometimes this guidance is not exactly the same as the information within the SmPC. But the first paragraph of chapter 4 of the green book says:
Recommendations on immunisation procedures are based on currently available evidence and experience of best practice. In some circumstances, this advice may differ from that in vaccine manufacturers’ Summaries of Product Characteristics (SPCs). When this occurs, the recommendations in this book (which are based on current expert advice received from the Joint
Committee on Vaccination and Immunisation JCVI) should be followed.
As a result we would follow the Green Book advice for a hepatitis schedule A vaccine. Chapter 17 page 9 says:
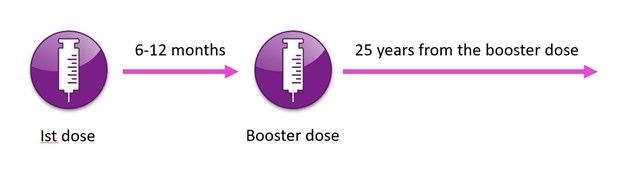
A single dose of monovalent hepatitis A vaccine provides protection for up to 12 months. However, for long term protection a second dose of hepatitis A vaccine should be given at 6 to 12 months after the initial dose. This second dose results in a substantial increase in the antibody titre and will give immunity for at least 25 years.
A number of studies have been carried out to examine the persistence of antibodies to hepatitis A after immunisation. Two studies aimed to assess the immunogenicity against hepatitis A in adults with a 2-dose primary vaccination (0, 6 months or 0, 12 months schedule) using a monovalent, inactivated hepatitis A vaccine, and indicated that both immunisation regimens resulted in persistence of vaccine-induced antibodies against HAV for at least 17 years after primary immunisation. Model-based estimates are consistent with estimates of seropositivity rates of up to 95% for at least 25 years. Until further evidence is available, reinforcing immunisation with a booster 25 years after a completed hepatitis A vaccine course with standard dose is therefore generally not needed except for those at ongoing risk or post-exposure to a person with hepatitis A. (See the Green Book chapter on page 9 to see this information which includes the references).
The image below is my attempt to explain it in a diagram!
CONCLUSION
- Give 2 doses of hepatitis A vaccine 6-12 months apart
- This information would apply to all the above vaccines
- The traveller then has 25 years of protection from the DATE of the COMPLETING DOSE of the course (which includes the paediatric vaccines)
- If the traveller did not attend for the booster dose on time, you would not restart the course but boost when they turn up (see below *)
- This length of protection is taken to apply to ALL hepatitis A products discussed in the Green book once a course of hepatitis A vaccine is given (including combination vaccines which have hepatitis A in them!)
- If a traveller seeks advice and it’s more than 25 years since their initial dose, you would give a further dose if your pre travel risk assessment indicates they will be at ongoing risk. Deciding this ‘ongoing risk’ is perhaps the challenging aspect and some examples discussed in case studies can be found in the Nuggets of Knowledge – Hepatitis A Vaccine (see below)
Other rules for hepatitis A vaccine include
- Monovalent hepatitis A vaccines are interchangeable, although it’s always best to complete with the brand you started with
- Always use an age appropriate vaccine (ie. one licensed for that specific age)
- Hepatitis A vaccine is always an NHS vaccine in an NHS setting
*Booster doses – in chapter 11 page 4 it says:
“where any course of immunisation is interrupted, there is normally no need to start the course again – it should simply be resumed and
completed as soon as possible”
Don’t forget, for a short e learning programme to reinforce all the above facts go to NUGGETS OF KNOWLEDGE – HEPATITIS A VACCINES
COMBINATION HEPATITIS A + B vaccines (from the hepatitis A perspective)
There are three relevant vaccines – Twinrix Adult, Twinrix paediatric, Ambirix.
- The content of hepatitis A in Twinrix Adult (720 ELISA units) is half the dose of hepatitis A that would be found in Havrix Monodose (1440 ELISA units)
- The content of hepatitis A in Twinrix Paediatric (360 ELISA units) is half the dose of hepatitis A that would be found in Havrix Junior Monodose (720 ELISA units)
- Ambirix is also a paediatric vaccine (used age 1-15) but the amount of hepatitis A in it is equivalent to a Havrix Junior Monodose (720 ELISA units)
Many errors occur in regard to the combined hepatitis A and B vaccines. Once the content and schedules are understood, there is nothing particularly difficult about their use. Follow these additional guidelines as well to ensure mistakes aren’t made when using twinrix adult or paediatric:
1. If giving a course of Twinrix you MUST ensure you give two doses of the vaccine prior to departure to obtain maximum protection for hepatitis A
2. If you start a course of hep A and hep B protection using either Twinrix Adult or Twinrix Paediatric vaccines then COMPLETE the course using these vaccines.
3. If you start a course of hep A and B protection using monovalent hep A and hep B vaccines, then you cannot use a combined hep A and hep B vaccine to complete the course. (this is all because of the content value of hepatitis A).
More information regarding FAQs on Twinrix is also found in the FAQ document below.
VERY USEFUL RESOURCES
NaTHNaC Hepatitis A factsheet from TravelHealthPro
Green Book Chapter 17 on Hepatitis A
Vaccine chart with all the current schedules include those for hepatitis A – see item no. 3 here




